Introduction
Breast cancer mortality has dropped since 1990, and this may be attributed to early detection through screening mammography and improved local regional therapies including surgery and radiotherapy. In turn, adjuvant systemic therapies, such as chemotherapy and/or endocrine therapy, are administered after surgery to eradicate potential micrometastatic disease1; these reduce recurrence and disease-related mortality rates in breast cancer patients2. On the other hand, systemic therapy can also be administered before surgery and is referred to as neoadjuvant or preoperative therapy, which has traditionally been used for locally advanced (clinical stage T3N1-N3M0) and inflammatory breast cancer (T4dN0-N3M0)3. At present, however, neoadjuvant chemotherapy (NACT) is also being used in earlier stages to induce a therapeutic response, reduce tumor size before surgery, and thus allow a shift from mastectomy to breast-conserving surgery, while also reducing the likelihood of axillary surgery4.
NACT was first applied by Portelles et al. to promote breast-conserving surgery, prolong overall survival, reduce tumor size, prevent ipsilateral recurrence, and improve breast cosmesis5.
Tumor response to NACT may also provide prognostic information. Achievement of a pathological complete response (pCR) after completion of NACT and surgical resection is associated with improved disease-free survival. This correlation depends to some extent on molecular subtype and is strongest for patients with triple-negative breast cancer (estrogen receptor -negative, progesterone receptor [PR]-negative, and human epidermal growth factor receptor 2 [HER2]-negative) with a pCR rate of 31%, and for HER2-positive breast cancer with a rate of 39%4 – both higher than the overall pCR rate of 19% for all patients5. In contrast, for hormone receptor-positive breast cancer, the pCR rate in response to NACT is lower (8%)6.
Studies on NACT have employed a variety of methods to assess tumor response; however, currently there are no standardized methods for its evaluation1.
Imaging modalities
Current methods to assess tumor response to NACT consist of physical examination and conventional breast imaging with mammography and ultrasound4. The accuracy of clinical breast examination to determine pCR in locally advanced breast cancer after NACT or endocrine therapy is 57%, which is lower than mammography (74%) and ultrasound (79%). Challenges with physical examination include the presence of firm fibroglandular tissue and post-therapy fibrosis, which can overestimate the amount of residual disease. Likewise, loss of interval palpability after treatment does not exclude residual tumor5.
Conventional breast imaging is performed before initiation of neoadjuvant therapy6. Contrast-enhanced magnetic resonance imaging (MRI), including diffusion-weighted imaging, has been considered more accurate for assessing pre- and post- NACT in breast cancer and for differentiating chemotherapy-induced fibrosis. Compared with MRI, mammography and ultrasound are considered suboptimal in such evaluations; however, MRI is more expensive, time-consuming, and less available. In our setting, primary imaging modalities – mammography and ultrasound – are increasingly used in combination to predict pCR4.
Diagnostic mammography should include craniocaudal, mediolateral, and full-field mediolateral views, with spot compression or magnification images at the site of the lesion, as well as contralateral full-field mediolateral oblique views7. Ultrasound should be performed for malignant breast masses and axilla if neoadjuvant therapy is planned4. Pretherapy imaging aims to determine the extent of disease for local-regional staging and to evaluate the contralateral breast. It is also important to confirm proper placement of the biopsy marker clip within the breast tumor and axillary lymph node (if sampled) before initiation of NACT, in case, a complete imaging response is achieved6.
Response evaluation criteria in solid tumors (RECIST)
Tumors respond differently to immunotherapies compared with chemotherapeutic drugs, highlighting the importance of assessing changes in tumor burden8.
The RECISTs were developed and published in 2000, based on the original World Health Organization guidelines first published in 1981. In 2009, revisions (RECIST 1.1) were introduced, incorporating changes such as a reduction in the number of lesions to evaluate, a new classification of lymph nodes as pathologic or normal, updated definitions of complete response (CR) and partial response (RP), and new methodologies for adequately measuring progression8 (Table 1).
Table 1. Summary of conventional tumor response criteria and modified strategies for immune response evaluations
| Conventional tumor response criteria | Measure | Partial response criterion | Disease progression criterion | Confirmation of disease progression | New lesion |
|---|---|---|---|---|---|
| WHO (1979) | Bi-dimensional (LD × LPD) | ≥ 50% reduction | ≥ 25% increase, new lesion, or disease progression in non-target | Not required | Defines disease progression |
| RECIST 1.0 (2000) | Unidimensional (LD) | ≥ 30% reduction | ≥ 20% increase, new lesion, or disease progression in non-target | Not required | Defines disease progression |
| RECIST 1.1 (2009) | Unidimensional (LD for non-nodal lesion and LPD for lymph nodes) | ≥ 30% reduction | ≥ 20% and ≥ 5 mm increase, new lesion, or disease progression in non-target | Not required | Defines disease progression |
|
LD: longest diameter; LPD: longest perpendicular diameter; PD: progressive disease; PR: partial response; RECIST: response evaluation criteria in solid tumors; WHO: World Health Organization. |
|||||
In view of ongoing controversies in the management and precise assessment of response to NACT, this study was designed with the objective of evaluating the concordance between mammography and ultrasound with histopathology to identify tumor response to neoadjuvant systemic therapy in breast cancer.
Material and methods
We conducted an observational, longitudinal, and retrospective cohort study from a database obtained from the Picture Archiving and Communication System of Fundación contra el Cáncer de Mama (FUCAM) (Mexico City, Mexico) including 1061 patients. A total of 52 patients met the inclusion criteria: patients who underwent neoadjuvant therapy, who had both mammography and ultrasound performed at the institution before and after initiation of neoadjuvant therapy, who underwent breast-conserving surgery or mastectomy, and who had histopathology reports available. Patients who did not complete the neoadjuvant regimen or who lacked both pre- and post-treatment imaging were excluded. Radiology reports were reviewed and compared with histopathology reports from the pathology department, considered the gold standard, to evaluate radiopathologic correlation. Post-NACT imaging features assessed included architectural distortion, presence of mass, calcifications, cystic degeneration, and absence of lesions. Treatment response was evaluated by measuring lesion dimensions along two major axes, independently for pathology samples and imaging studies. Evaluation also included tumor size at resection and histologic classification of chemotherapy response as complete, minimal residual, moderate residual, or extensive residual (Residual cancer burden [RCB] criteria). Statistical analysis included percentages, mean, standard deviation, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and Cohen’s kappa index to assess concordance between post-NACT imaging and final surgical histopathology. P < 0.05 was considered statistically significant.
Results
From the initial database of 1061 patients, 52 met the inclusion criteria, with a mean age of 50.9 years. The most frequent findings were left breast involvement (55.8%), heterogeneously dense breast tissue (73.1%), luminal B phenotype (34.6%), followed by triple-negative (28.8%), and poorly differentiated histology (71.2%). The most common imaging feature after NACT was mass/solid nodule (51.9%), while the least frequent were calcifications and cystic degeneration (1.9% each) (Table 2).
Table 2. Description of imaging and histopathological findings
| Variable | n | % | |||
|---|---|---|---|---|---|
| 52 | 100 | ||||
| Affected side | |||||
| Right | 20 | 38.5 | |||
| Left | 29 | 55.8 | |||
| Bilateral | 3 | 5.8 | |||
| Breast density | |||||
| Heterogeneously dense | 38 | 73.1 | |||
| Scattered fibroglandular | 13 | 25.0 | |||
| Extremely dense | 1 | 1.9 | |||
| Imaging findings | |||||
| Nodule | 27 | 51.9 | |||
| Architectural distortion | 17 | 32.7 | |||
| Calcifications | 1 | 1.9 | |||
| Cystic degeneration | 1 | 1.9 | |||
| No lesion | 6 | 11.5 | |||
| Phenotype | |||||
| Luminal B | 18 | 34.6 | |||
| Triple-negative | 15 | 28.8 | |||
| Pure HER2 | 13 | 25.0 | |||
| Luminal + HER2 | 4 | 7.7 | |||
| Luminal A | 2 | 3.8 | |||
| Histological grade | |||||
| Moderately differentiated | 15 | 28.8 | |||
| Poorly differentiated | 37 | 71.2 | |||
|
The values in bold correspond to the most frequent findings. HER2: human epidermal growth factor receptor 2. |
|||||
Pathologic response by imaging was classified as partial in 55.8% of cases (Figs. 1 and 2), complete in 34.6% (Figs. 3 and 4), stable (Fig. 5), or progressive disease in 9.6%. By pathology (RCB), response was complete in 53.8%, partial in 34.6%, and progressive in 11.5% (Fig. 6).
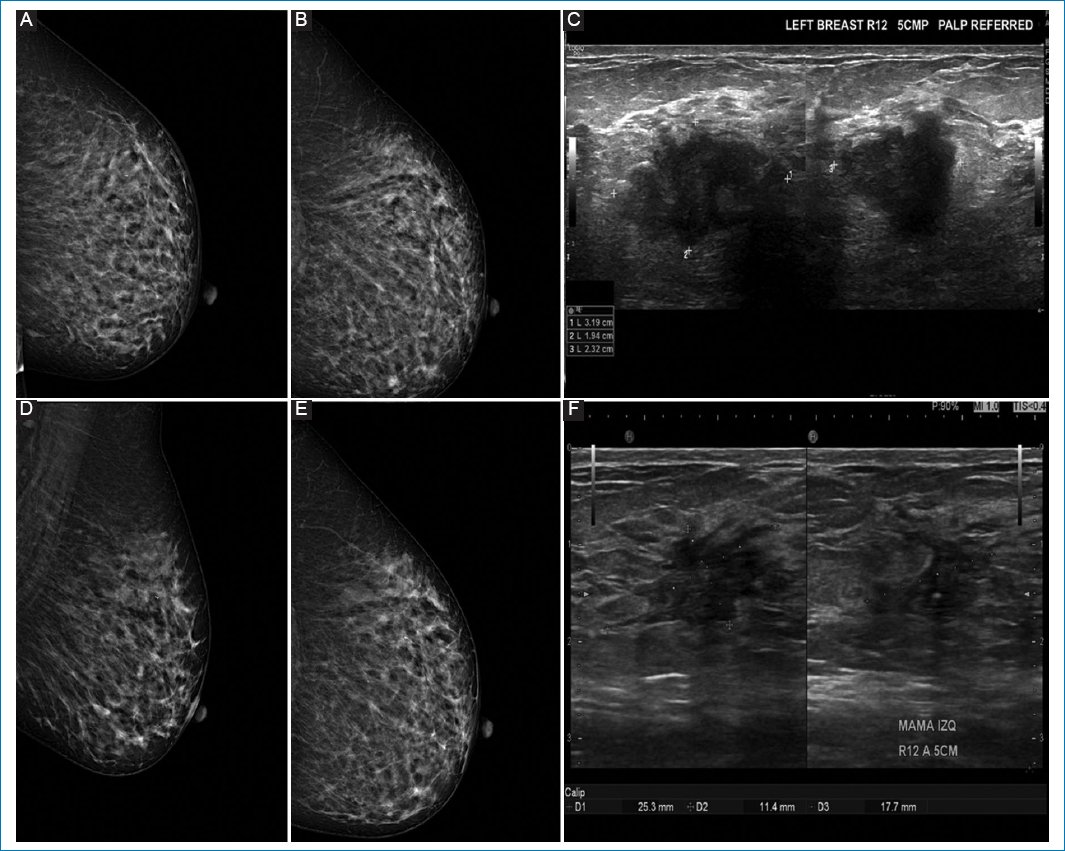
Figure 1. A-C: pre-neoadjuvant images. D-F: post-neoadjuvant images. 51-year-old woman diagnosed with left breast cancer, clinical stage IIA by T2N0M0, Luminal B, estrogen receptor 60%, PR 90%, with human epidermal growth factor receptor 2 overexpressed, Ki67 45%. A and B: pre-neoadjuvant mastography specimen, with irregular nodule, in upper outer quadrant. C: ultrasound same patient with irregular nodule in R12 at 50 mm, from the nipple. D and E: mastography. F: ultrasound showing a reduction > 20%, which corresponds to partial response.
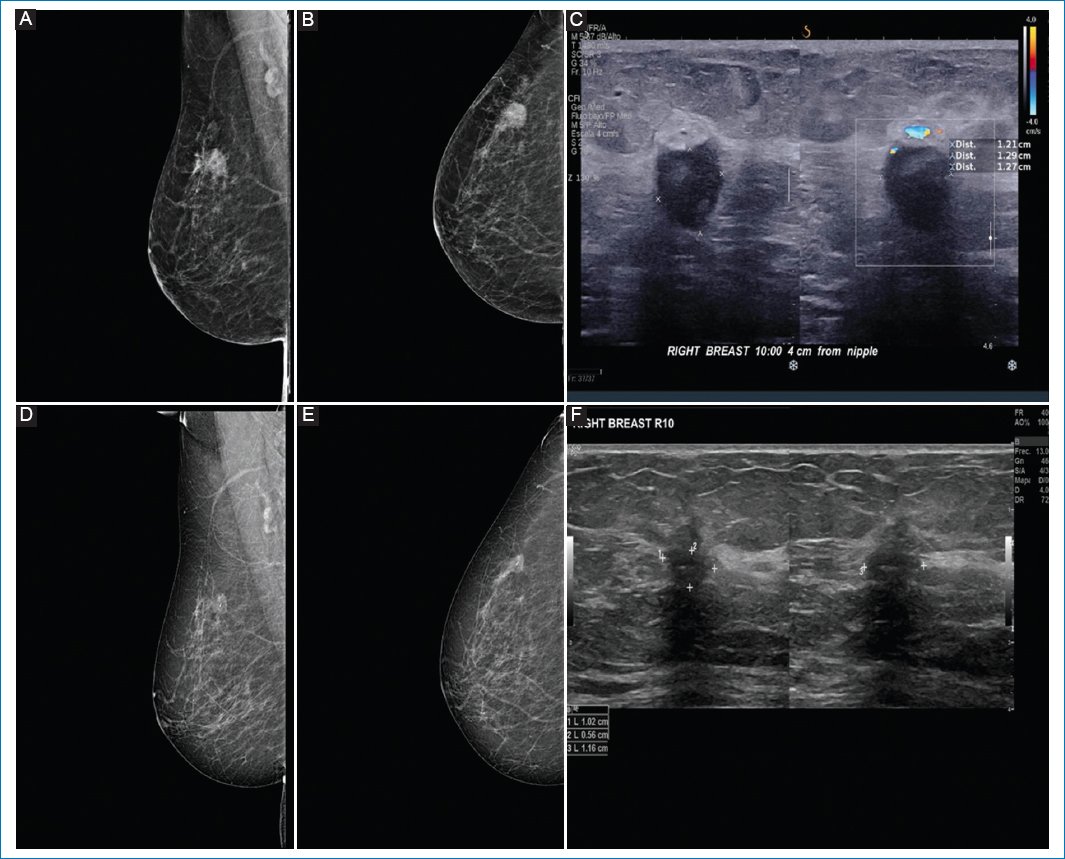
Figure 2. A-C: pre-neoadjuvant images. D-F: post-neoadjuvant images. 64-year-old patient with diagnosis of infiltrating ductal carcinoma of the right breast, clinical stage IIB, triple negative. A and B: pre-neoadjuvant mastography specimen with irregular nodule in upper outer quadrant. C: ultrasound specimen with irregular nodule in R10. D and E: correspond to the mastography. F: ultrasound showing a nodule with partial response to treatment and the presence of intratumoral clip.
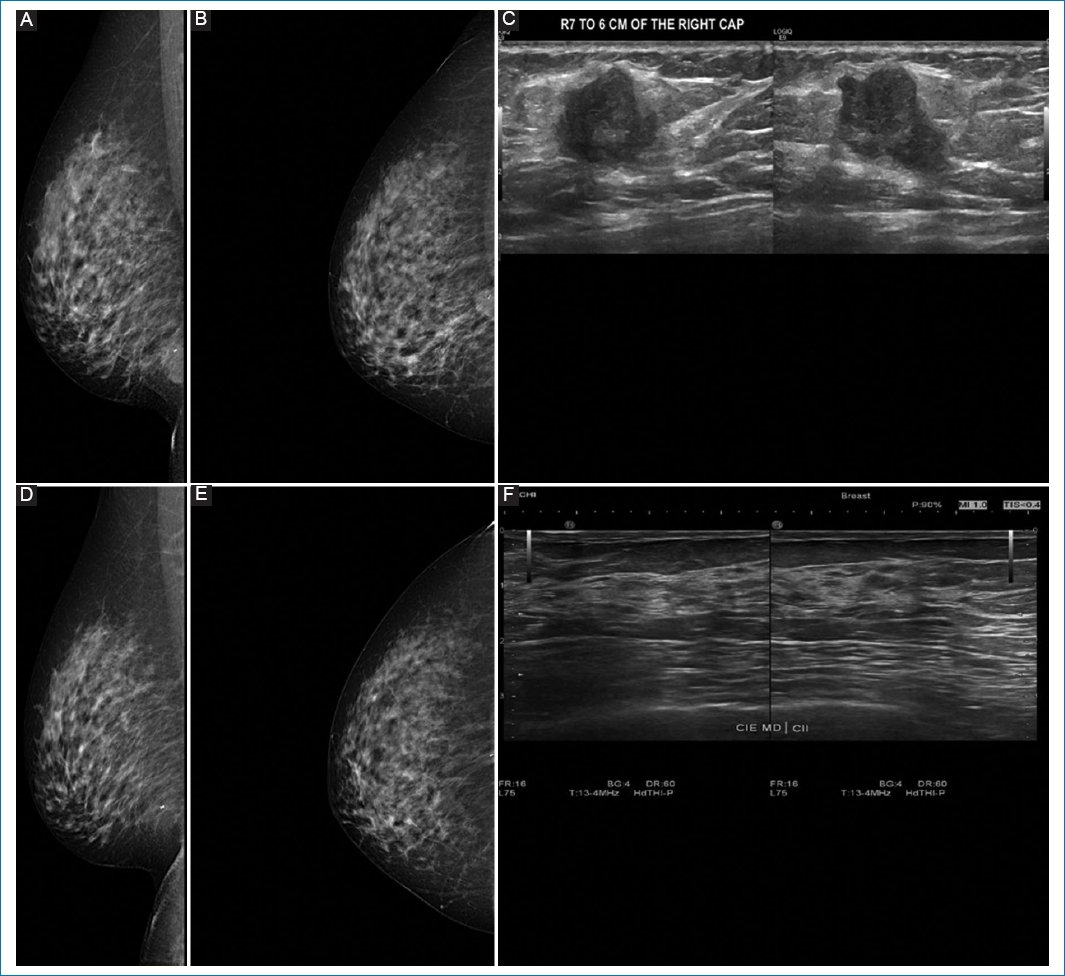
Figure 3. A-C: pre-neoadjuvant images. D-F: post-neoadjuvant images. 41-year-old woman diagnosed with right breast cancer, clinical stage IB by T1cN0M0, Luminal B with low hormone receptor expression, estrogen receptor 0%, PR 5%, human epidermal growth factor receptor 2 not amplified (Silver in situ hybridization) KI67 80%. A and B: shows pre-neoadjuvant mastography, with irregular nodule, in lower inner quadrant, showing intratumoral clip. C: ultrasound same patient with irregular nodule in R7 at 60 mm from the nipple. D and E: show mastography with clip, no nodule is visualized. F: breast ultrasound in R7 at 60 mm from the nipple, without presence of nodule. Images correspond to complete pathologic response corroborated by pathology.
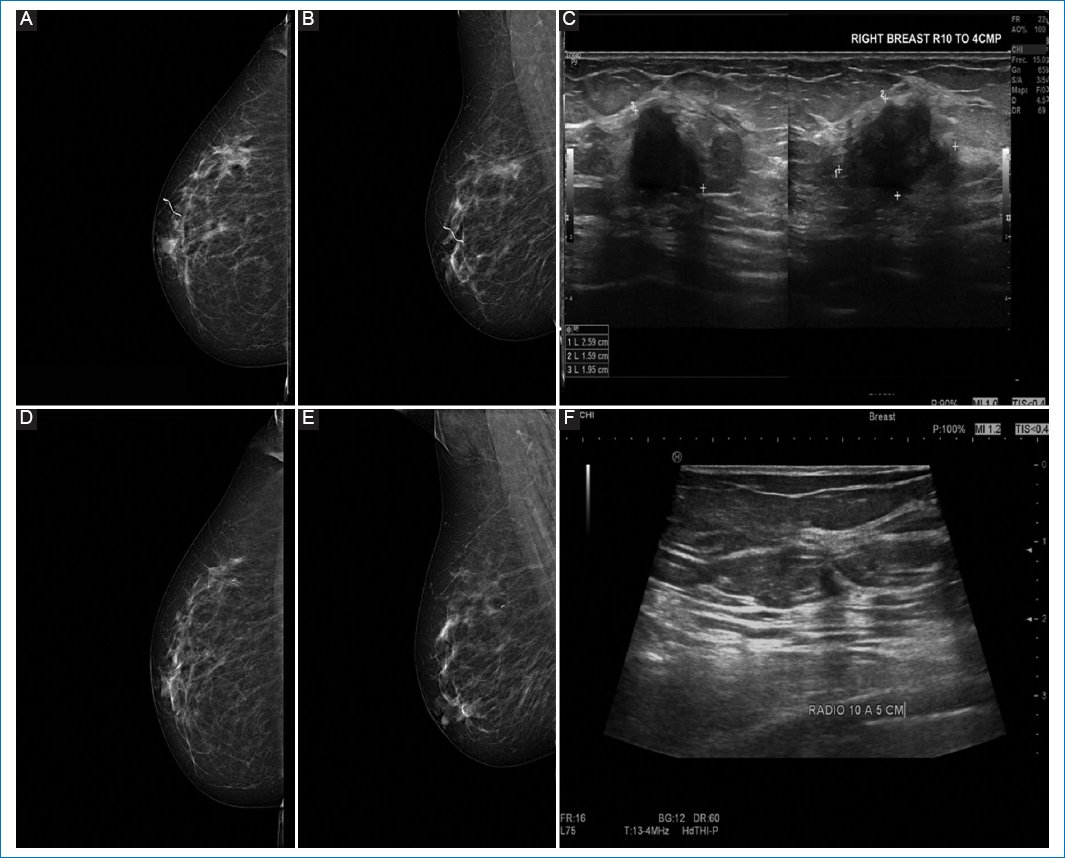
Figure 4. A-C: pre-neoadjuvant images. D-F: post-neoadjuvant images. 56-year-old female, diagnosed with infiltrating ductal carcinoma, grade II in right breast. Clinical stage IIA by T2 N0 M0, estrogen receptor 0%, PR 0%, human epidermal growth factor receptor 2 negative, and KI 67 5%. A and B: preneoadjuvant mastography specimen, with irregular nodule in the upper outer quadrant. C: ultrasound specimen of the same patient, with irregular nodule in R10 at 50 mm from the nipple. D and E: correspond to the mastography. F: ultrasound showing only clip, which corresponds to complete response.
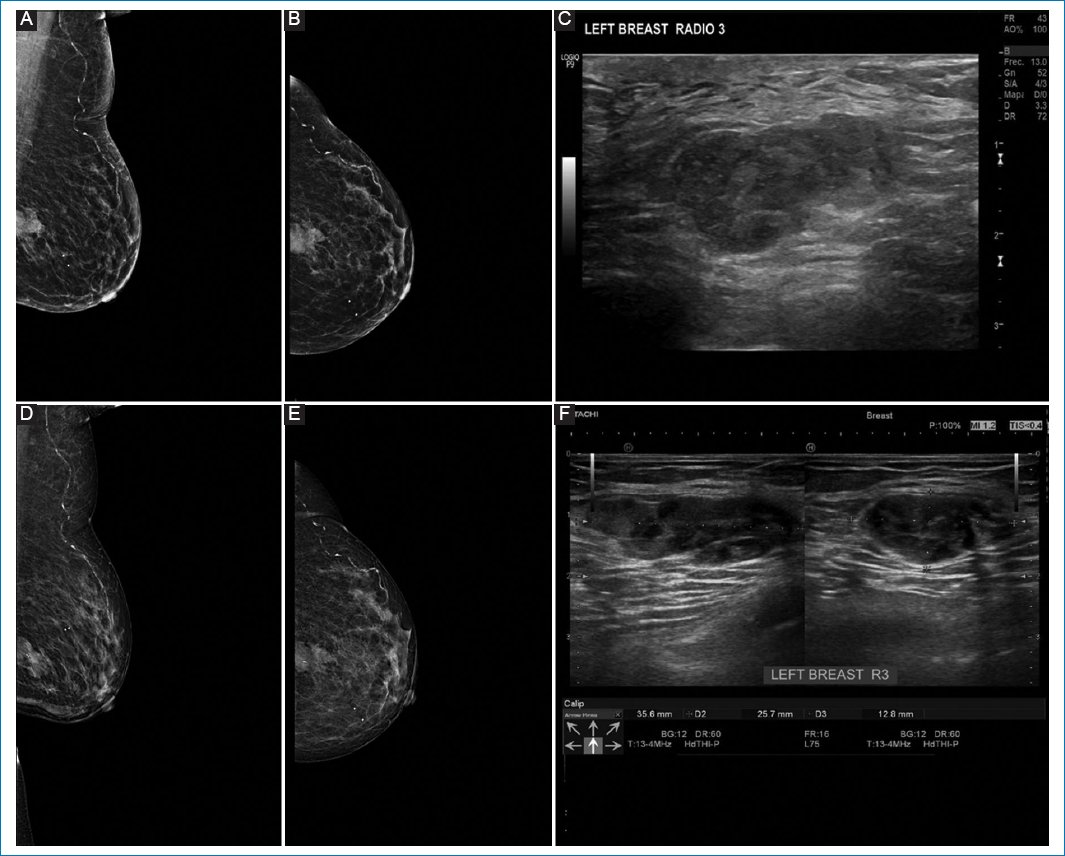
Figure 5. A-C: pre-neoadjuvant images. D-F: post-neoadjuvant images. 78-year-old patient with a diagnosis of infiltrating ductal carcinoma, with mucinous differentiation, moderately differentiated grade II of the left breast, clinical stage IIA, Luminal B. A and B: preneoadjuvant mastography specimen, with irregular microlobulated nodule, in the interline of outer quadrants, posterior third. C: ultrasound specimen with irregular nodule in R3. D and E: correspond to the mastography. F: ultrasound shows nodule without significant changes with respect to the previous one, in relation to stable disease.
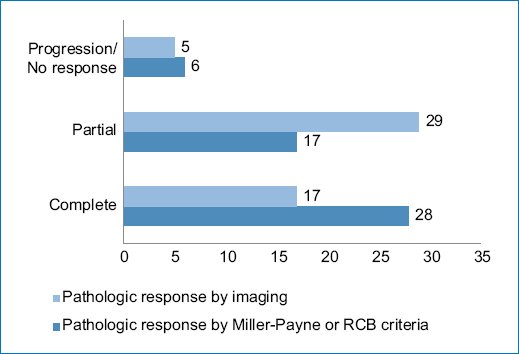
Figure 6. Radiopathologic response according to the type of response found.
In the evaluation of response to NACT, concordance was calculated between conventional imaging studies (mammography and ultrasound) and histopathological results. For CR, the combination of mammography and ultrasound achieved a sensitivity of 64%, specificity of 100%, PPV of 100%, and NPV of 71%, with good concordance (κ = 0.62; 95% confidence interval [CI], 0.38-0.87). For partial response, sensitivity was 94%, specificity 65%, PPV 59%, and NPV 96%, with moderate concordance (κ = 0.52; 95% CI, 0.28-0.75). For disease progression, sensitivity was 67%, specificity 98%, PPV 80%, and NPV 96%, with good concordance (κ = 0.69; 95% CI, 0.43-0.96). Therefore, overall, when evaluating both partial and CRs, sensitivity was 98%, specificity 67%, PPV 96%, and NPV 80%, with good concordance (κ = 0.70; 95% CI, 0.43-0.96) (Table 3).
Table 3. Concordance and diagnostic accuracy of mammography and ultrasound findings with histopathology report
| Variable | Pathological response Miller Payne or Residual cancer burden | Sensitivity | Specificity | Positive predictive value | Negative predictive value | Kappa* | 95% confidence interval | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Complete | Partial | Progression | Total | |||||||
| Pathological response by imaging | ||||||||||
| Complete | 18 | 0 | 0 | 18 | 64 | 100 | 100 | 71 | 0.62 | 0.38-0.87 |
| Partial | 10 | 17 | 2 | 29 | 94 | 65 | 59 | 96 | 0.52 | 0.28-0.75 |
| Progression | 0 | 1 | 4 | 5 | 67 | 98 | 80 | 96 | 0.69 | 0.43-0.96 |
| Total | 28 | 18 | 6 | 52 | 98 | 67 | 96 | 80 | 0.70 | 0.43-0.96 |
|
* Weighted kappa index. |
||||||||||
Discussion
Patients on NACT as first-line therapy, followed by definitive surgery (breast-conserving or mastectomy), were evaluated. Results show that conventional breast imaging is useful for assessing response to NACT, with adequate sensitivity and specificity for identifying partial or CR and disease progression. Concordance was higher for detecting CR and progression, while lower for partial response, consistent with data reported by Makanjuola et al.2.
One major limitation of this study was the absence of a standardized imaging protocol for patients undergoing NACT, requiring both pre- and post-treatment imaging, which reduced the eligible sample to 52 patients (4.9% of the 1061 treated during the study period).
At present, radiologists are responsible for evaluating response in patients receiving NACT through ultrasound, mammography, or MRI. However, Breast Imaging-Reporting and Data System (BI-RADS) classification lacks a specific category for post-NACT radiologic response, which may result in inadequate lexicon or discordance during evaluation. Accurate identification of response is now imperative, as it influences definitive surgical approach, shifting from mastectomy to breast-conserving surgery. Some trials have reported up to 83% pCR in HER2-positive, hormone receptor-negative cancers, according to Rea et al.9.
The use of BI-RADS category 6 for a radiologically visible mass with only a clip remaining creates confusion, as two distinct findings share the same coding. It is suggested to maintain BI-RADS 6, which preserves the cancer status confirmed by biopsy, whereas “zero” or “null” would indicate complete resolution of a known tumor. Consistent with Makanjuola et al.2, a special BI-RADS category should be assigned for this reference point, promoting precise radiologic reporting, guiding preparation for surgical resection or vacuum biopsy, and providing more clinically useful radiopathologic correlation.
Conclusion
Assessment of pathological response after NACT with conventional methods has shown greater sensitivity and specificity when combined. However, mammography has limitations in dense breasts (categories C-D), where ultrasound is usually more sensitive. Thus, mammography and ultrasound together can accurately predict treatment response in patients undergoing NACT, as accessible imaging methods. Nonetheless, further research with standardized protocols is needed to confirm these results and to provide more efficient and effective diagnostic methods for response evaluation.
Acknowledgments
The authors would like to thank the radiologists, residents, and staff of the Imaging Department of Fundación contra el Cáncer de Mama (FUCAM) for facilitating the study and providing the information.
Funding
The authors declare that they have not received funding.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical considerations
Protection of humans and animals. The authors declare that no experiments involving humans or animals were conducted for this research.
Confidentiality, informed consent, and ethical approval. The authors have obtained approval from the Ethics Committee for the analysis of routinely obtained and anonymized clinical data, so informed consent was not necessary. Relevant guidelines were followed.
Declaration on the use of artificial intelligence. The authors declare that no generative artificial intelligence was used in the writing of this manuscript.
