Introduction
Tetralogy of Fallot (TF) is the most common cyanotic congenital cardiopathy, representing around 5% of all heart diseases of congenital origin. It was first described in 1671 by Dan Stentson, but until 1888, Doctor Etienne-Louis Fallot detailed the four main cardiac defects derived from malformation of the aorticopulmonary septum1. Pathophysiologically, these defects cause the development of a right-to-left shunt and right ventricular (RV) outflow tract obstruction whose main clinical manifestations are generalized cyanosis, recurrent respiratory distress, and heart failure2.
To improve the symptomatology, the treatment consists of surgical repair of the septal defects during the early months or years of life. Although the post-operative survival rate at 5 and 10 years is 92 and 74%, respectively, up to 14% of cases require a second intervention in the medium or long term due to complications such as arrhythmias and heart failure depending on initial anatomy, surgical approach, and post-operative control and follow-up over time1.
Repaired TF associated heart failure is not only the main long-term complication and primary admission diagnosis, but also the major cause of death among these patients due to an increased risk of readmission with a higher rate of mortality. This is not only clinically relevant due to the need of treating complications in specialized cardiovascular centers, but also has a financial impact associated with out-of-pocket expenses for medical attention3.
Therefore, we report the case of a 73-year-old female patient with corrected TF who arrived at our cardiology service for diagnostic evaluation and therapeutic management decades after undergoing corrective surgery. Even though the first treated surgically TF was in 1945, with a palliative subclavian–pulmonary arterial shunt4, our patient was treated with transannular patches successfully.
Case presentation
A 73-year-old woman arrives at the emergency department presenting dyspnea and heart failure symptoms with moderate exertion for 2 weeks, worsening of her symptoms, developing heart failure with moderate exertion accompanied with a history of falls from her support plane 4 times with syncope. As her symptoms persisted, she decided to go to the emergency department to manage her current condition.
She arrives with disorientation in time and place, general weakness, and anterograde amnesia. She has a history of hypertension; decompensated heart failure New York Heart Association class IV/American Heart Association stage C (NYHA IV/AHA C); chronic obstructive pulmonary disease Global Initiative for Chroninc Obstructive Lung Disease Stage D (GOLD D), and a TF corrected surgically in childhood. On auscultation, a holosystolic murmur predominantly in the aortic area increases during the Rivero-Carvallo maneuver.
In the examination, there was a remarkably generalized pitting edema of 1 year of evolution Godett 4+, with hot and painful erythema in the lower hemiabdomen and lower limbs of 2 weeks of evolution, and peribuccal cyanosis. The pulse rate was 65 beats/min; blood pressure of 137/75; respiratory rate of 16/min, and oxygen saturation of 75%. Complementary diagnostic studies were performed to complete the clinical assesment (Fig. 1). The electrocardiogram showed a non-sinus rhythm (Fig. 2), with a right bundle branch block and atrial tachycardia with a 3rd grade atrioventricular (AV) block, with non-specific intraventricular conduction alterations and secondary ventricular alterations. The echocardiogram showed chronic changes with cardiac remodeling characterized by a remarkable dilatation of both atria and right ventricle, and a hypertrophic interventricular septum.
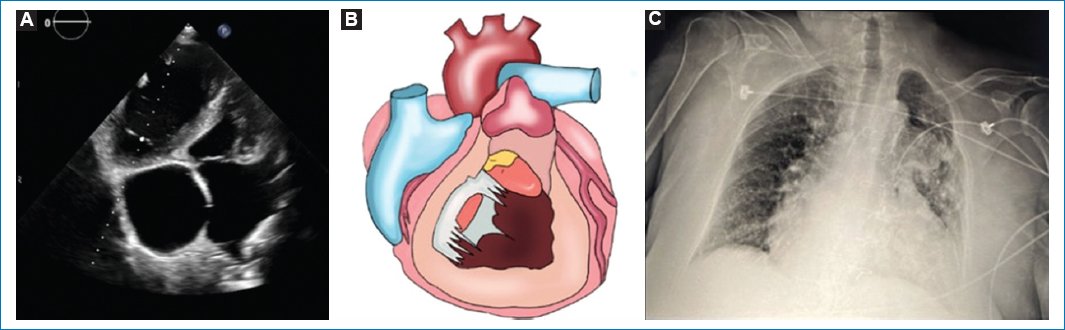
Figure 1. A: four-chamber view on transthoracic echocardiography: showing dilation of both atria, atrial septal defect, hypertrophic interventricular septum, and a small left ventricle chamber due to right ventricle dilation. B: illustration of cardiac remodeling due to congenital heart disease. C: radiography with data of pulmonary congestion and cardiomegaly.
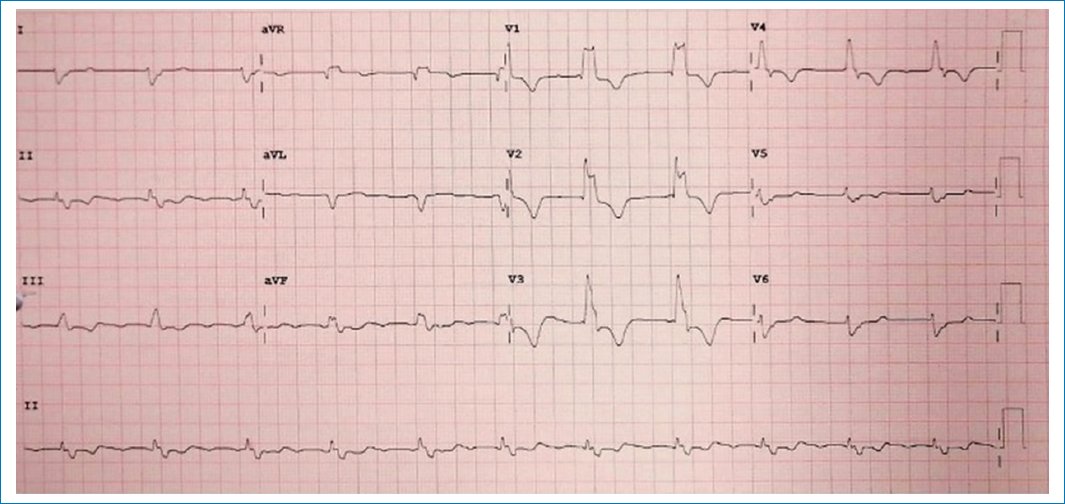
Figure 2. Patient’s electrocardiogram at the emergency department. 12-lead electrocardiogram with standard calibration, non-sinus rhythm, with right bundle branch block and junctional rhythm, heart rate of 65 bpm, upright position, and levorotatory transition. With non-specific intraventricular conduction disturbances and secondary ventricular repolarization disturbances.
The initial approach was with loop diuretics to reduce pulmonary congestion and peripheral edema. The patient was discharged with clinical improvement and with optimal treatment for heart failure.
Discussion
TF is a congenital cardiac disease characterized by underdevelopment of the RV infundibulum, with an anterior and leftward displacement of the interventricular septum, and associated with right ventricle outflow pulmonary stenosis and large ventricular septal defect (VSD). The first description was done by Roger, and Wilson in 19085. A second case was not reported until Kurtz et al. described their case in 19276. TF was first treated surgically by Blalock and Taussig in 1945, with a palliative subclavian-pulmonary arterial shunt4. Other types of systemic-Potts et al. introduced pulmonary arterial shunts in 19467, Klinner in 19618, Davidson in 19559, and palliation by direct relief of pulmonary stenosis with a closed technique was introduced by Holmes Sellors10 and Brock11,12 in 1948. TF was first successfully repaired by Lillehei et al. at the University of Minnesota in 1954 using controlled cross-circulation with another person serving as oxygenator13. The first successful repair of TF using a pump-oxygenator was done by Kirklin et al. at the Mayo Clinic in 195514. Cohen et al. introduced patch enlargement of the RV infundibulum in 195715, and Kirklin et al. reported in 1959, the use of transannular patches16. Use of an RV-pulmonary trunk conduit for TF with pulmonary atresia was done by Kirklin et al. in 196517, and Ross and Somerville first reported the use of a valved extracardiac conduit for this purpose in 196618.
TF is one of the most common cardiac malformations19. Its prevalence mainly consists of 3 cases/10,000 live births, and it accounts for about 5-10% of all congenital defects, with substantial geographic variations. It seems to be sporadically associated with microdeletions on 22q, highly related to retinoic acid intake and untreated diabetes in pregnancy, and with a risk of recurrence of about 3% in siblings20,21. It consists of four main malformations: interventricular communication, a narrowing of the pulmonary valve and main pulmonary artery, an enlargement of the aortic valve, and RV hypertrophy20,22. The severity of the disease depends on the amount of outflow obstruction of the right ventricle. Clinically, this pathology usually manifests during the neonatal stage with generalized cyanosis, conditioned by the presence of a right-to-left shunt, leading to hypoxemia and severe cyanosis19. For the diagnosis, echocardiography is the gold standard to evaluate the VSD, the obstruction of the RV outflow tract, pulmonary valve, and pulmonary arteries pressure, the ventricular function and size, and if there is any tricuspid regurgitation and increase of RV pressure23.
Regarding treatment, surgical management is the alternative of choice. Patient age and surgical intervention are determinants of survival in adults; of the patients in their 30’s, 90% survive with a surgical repair, while only 10% survive without surgical intervention3,19. Therefore, it is crucial to make a timely diagnosis and perform early surgical management. Years after a surgical repair, the main hemodynamic consequences are pulmonary regurgitation and a degree of residual RV outflow obstruction, especially as patients age. Evidence suggests that both entities raise gradual regression of right ventricle volume to near pre-operative values after 10 years of replacement surgery, therefore, it leads to pulmonary heart disease, also called “Cor pulmonale”, with heart remodeling by chronic volume or pressure overload in the right ventricle1,24. Moreover, this cascade of events leads to myocardial damage, dilatation of cavities, electrical conductions disorders, and changes in ventricular mechanics with important clinical implications which eventually manifest with heart failure as observed in this case1,22.
In this context, authors debate that follow-up evaluation with imaging studies like echocardiography and magnetic resonance imaging continues to be a crucial element in planning the approach of these patients to address the problem in time, not so prematurely as to lead to recurrent surgical procedures, but not so late as to reach stages of major deterioration in which clinical and financial impact grows exponentially with an increased risk of readmission, twice the cost of hospitalization compared to non-congenital heart failure, an 19-fold higher risk of cardiovascular events and a higher rate of morbimortality1,3,22. Highlighting the need for adequate follow-up to prevent complications, especially in a country where there are few specialized centers or where they are so centralized that access to them is difficult25.
Conclusion
The TF is more prevalent during childhood than adulthood and has a survival rate until the fourth or fifth decade of life if it is surgically corrected on time. This case was an unusual presentation of a corrected TF since the patient arrived with signs and symptoms of decompensated pulmonary heart disease, including cardiac remodeling on electrocardiogram and echocardiogram. It has been almost 40 years since the first published reports of primary repair in neonates and infants, and more than 15 years since one-stage and two-stage repair have been compared with formal analysis. Nevertheless, current practice varies among individual surgeons and institutions, with one- and two-stage repair frequently practiced using varying protocols. Therefore, the highlights of this case show us a successful surgical repairment and the importance of adequate medical follow-up to improve results, assess other therapeutic strategies, and have an impact on the financial and morbimortality evolution of patients.
Acknowledgments
The authors would like to thank the department of cardiovascular surgery of the Centro Médico ABC for their collaboration on the article.
Funding
The authors declare that they have not received funding.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical considerations
Protection of humans and animals. The authors declare that no experiments involving humans or animals were conducted for this research.
Confidentiality, informed consent, and ethical approval. The authors have followed their institution’s confidentiality protocols, obtained informed consent from patients, and received approval from the Ethics Committee. The SAGER guidelines were followed according to the nature of the study.
Declaration on the use of artificial intelligence. The authors declare that no generative artificial intelligence was used in the writing of this manuscript.