Introduction
The post-anesthetic recovery phase represents a criti cal moment in the perioperative period. During this stage, the residual effects of anesthetics and the physiological changes resulting from surgery converge. Within this unstable clinical environment, various adverse events may occur, ranging from acute postoperative pain or postoperative nausea and vomiting (PONV) to hypoxemia, arrhythmias, neurological deterioration, or cardiac arrest, all of which require immediate intervention1,2.
Some of these events are directly related to anesthesia, such as residual neuromuscular blockade, which predisposes to airway obstruction, or respiratory depression secondary to opioids, benzodiazepines, or anesthetics, particularly in vulnerable patients. Acute respiratory events may also occur, including laryngospasm, bronchospasm, aspiration, or significant hypoventilation, situations in which an immediate therapeutic plan must be initiated. In exceptional but clinically documented cases, the post-anesthesia care unit (PACU) may become the setting for cardiorespiratory arrest requiring advanced resuscitation2.
Despite these risks, the PACU is often underestimated within the perioperative setting and viewed as a “transition” space rather than a critical care environment. This perspective minimizes the fact that the PACU is, in reality, a short-stay intensive monitoring unit in which the patient’s physiological status has not yet stabilized and where any diagnostic delay may have deleterious consequences. Therefore, its operation must ensure trained personnel, including an anesthesiologist and nursing staff skilled in critical care, along with clear early detection protocols and discharge criteria based on guidelines that guarantee patient safety at all times – not only operational efficiency3–5.
Despite advances in monitoring, anesthetic techniques, and institutional protocols, adverse events in the PACU remain frequent and clinically relevant. Among the most widely reported in the literature are acute postoperative pain, PONV, episodes of hypertensive crises and hypotension, as well as respiratory complications (including hypoxemia, hypoventilation, upper airway obstruction, and less commonly laryngospasm or bronchospasm). These are accompanied by residual effects of neuromuscular blockers, hypothermia, shivering, arrhythmias, urinary retention, and episodes of agitation or postoperative delirium, all of which may compromise patient safety. Delayed detection or inadequate treatment may prolong recovery room stay, delay hospital discharge, and increase the need for unplanned interventions such as reintubation or transfer to intensive or intermediate care units.
In recent years, international organizations such as the American Society of Anesthesiologists (ASA) and The Joint Commission have emphasized the role of the PACU as a perioperative quality control point. National regulations, such as NOM-006-SSA3-2017, have also reinforced the need for structured monitoring and standardized discharge protocols. However, local evidence regarding incidence, clinical profile, and factors associated with adverse events in the PACU remains limited3–5.
Former studies have reported variable frequencies of immediate post-anesthetic complications: moderate to severe pain in up to 20-30%, PONV in 10-15%, and respiratory or cardiovascular events in 2-6%. Variability between centers reflects not only differences in patient and surgical populations but also disparities in definitions, documentation, and clinical response to these events6–10.
Systematic documentation of adverse events in the PACU enables identification of risk patterns, comparison of quality standards, design of targeted interventions, and ultimately improvement of anesthetic-surgical safety and outcomes. In this context, conducting observational studies using local data is essential to guide institutional policies and update recovery protocols.
Therefore, the objective of this study was to estimate the incidence of adverse events in the PACU at Centro Médico ABC and to analyze the clinical and anesthetic factors associated with their occurrence. Our aim is to provide useful evidence to guide prevention strategies, optimize resource allocation, and strengthen the quality of post-anesthetic care in our setting.
Methods
We conducted an observational, retrospective, cross-sectional study to estimate the frequency and types of complications in the PACU at Centro Médico ABC. A total of 2,025 consecutive patients undergoing surgery under any type of anesthesia and admitted to the PACUs of the Observatorio and Santa Fe campuses from February 1st, 2019 through June 30th, 2020, were included.
Data was collecte upon PACU admission using a structured form administered by anesthesiology residents. The following variables were recorded: date of procedure, age, sex, surgical specialty or type of surgery, preoperative ASA classification, relevant comorbidities (hypertension, diabetes, heart disease, COPD, among others), type of anesthesia (general, regional, or combined), and surgical duration in minutes.
The data collection form was developed by the research team based on clinical literature and institutional guidelines on post-anesthetic monitoring. Its content was reviewed by Centro Médico ABC Ethics Committee as part of protocol approval and by attending physicians from the Department of Anesthesiology involved in the study, all with clinical experience in PACU patient management. This review ensured content validity and clinical consistency of included items. Prior to formal implementation, the form was piloted in a phase not included in the final analysis, allowing minor wording adjustments and training of resident staff responsible for its administration. Although formal statistical reliability testing was not performed, the instrument was applied in a standardized and prospective manner throughout the study period.
PACU complications were defined a priori and considered present when at least one operational criterion, described in table 1, was met. Other complications (allergy, arrhythmias, cardiorespiratory arrest, acute urinary retention, etc.) were documented and validated by the responsible anesthesiologist.
Table 1. Baseline and perioperative characteristics of the cohort (n = 2,025). Continuous variables are expressed as median and IQR after normality was rejected; categorical variables as n (%). Surgical time in minutes
| Variable | Summary |
|---|---|
| Age | Median 50.0 (IQR, 35.0–64.0) (n = 2,025) |
| Surgical time | Median 150.0 (IQR, 120.0–180.0) (n = 2,025) |
| Regional block | Yes: 88 (4.3%). No: 1,937 (95.7%) |
| Smoker | Yes: 430 (21.2%). No: 1,595 (78.8%) |
| Elective surgery | Yes: 1,887 (93.2%). No: 138 (6.8%) |
| Oncologic | Yes: 215 (10.6%). No: 1,810 (89.4%) |
| COPD | Yes: 18 (0.9%). No: 2,007 (99.1%) |
| Pulmonary embolism | Yes: 12 (0.6%). No: 2,013 (99.4%) |
| OSA | Yes: 15 (0.7%). No: 2,010 (99.3%) |
| DM | Yes: 135 (6.7%). No: 1,890 (93.3%) |
| HTN | Yes: 395 (19.5%). No: 1,630 (80.5%) |
| Dyslipidemia | Yes: 111 (5.5%). No: 1,914 (94.5%) |
| Heart disease | Yes: 90 (4.4%). No: 1,935 (95.6%) |
| CKD | Yes: 20 (1.0%). No: 2,005 (99.0%) |
| Liver disease | Yes: 14 (0.7%). No: 2,011 (99.3%) |
| Neurologic | Yes: 82 (4.0%). No: 1,943 (96.0%) |
| Asthma | Yes: 24 (1.2%). No: 2,001 (98.8%) |
| Hypothyroidism | Yes: 164 (8.1%). No: 1,861 (91.9%) |
| Autoimmune diseases | Yes: 49 (2.4%). No: 1,976 (97.6%) |
| Coagulopathies | Yes: 6 (0.3%). No: 2,019 (99.7%) |
| Anxiety | Yes: 12 (0.6%). No: 2,013 (99.4%) |
| Depression | Yes: 21 (1.0%). No: 2,004 (99.0%) |
| Insulin resistance | Yes: 32 (1.6%). No: 1,993 (98.4%) |
| Obesity | Yes: 55 (2.7%). No: 1,970 (97.3%) |
| COVID | Yes: 6 (0.3%). No: 2,019 (99.7%) |
| Other | Yes: 232 (11.5%). No: 1,793 (88.5%) |
| Campus | Santa Fe: 1,459 (72.0%). Observatorio: 566 (28.0%) |
| Sex | Male: 1,027 (50.7%). Female: 998 (49.3%) |
| Type of surgery | General surgery: 693 (34.2%). Trauma and orthopedics: 448 (22.1%). Urology: 282 (13.9%). Neurosurgery: 185 (9.1%). Otolaryngology: 145 (7.2%). Plastic surgery: 125 (6.2%). Oncologic surgery: 83 (4.1%). Vascular: 21 (1.0%). Gynecology: 19 (0.9%). Thoracic: 17 (0.8%). Cardiology: 4 (0.2%). Maxillofacial: 3 (0.1%) |
| Technique | Balanced general anesthesia: 1,752 (86.5%). TIVA: 170 (8.4%). Combined: 88 (4.3%). Neuraxial: 9 (0.4%). Regional anesthesia: 6 (0.3%) |
| ASA | II: 1,139 (56.2%). I: 616 (30.4%). III: 254 (12.5%). IV: 15 (0.7%). V: 1 (0.0%) |
|
ASA: American Society of Anesthesiologists classification; COVID: coronavirus disease; DM: diabetes mellitus; HTN: systemic hypertension; CKD: chronic kidney disease; IQR: interquartile range; OSA: obstructive sleep apnea syndrome; TIVA: total intravenous anesthesia. | |
The primary endpoint of the study was the presence of at least 1 PACU complication (dichotomous variable). Secondary endpoints included PACU stay > 1 hour, occurrence of each complication separately, No. of complications per patient, PACU readmission, transfer to the intensive care unit, and reintervention within 24-48 hours.
Sample size was calculated to estimate proportions with a 2.5% margin of error and 95% confidence level, assuming p = 0.5 for maximum variance; the minimum required sample was 1,523 patients, and the observed sample (2,025) exceeded this threshold.
Statistical analysis was conducted at 3 levels. First, descriptive analysis: means and standard deviations or medians and interquartile ranges were reported for continuous variables according to distribution, and relative frequencies with 95% confidence intervals (95% CI) for categorical variables. Second, bivariate analysis: groups were compared using Student’s t-test or Mann-Whitney test, and chi-square or Fisher’s exact test as appropriate, exploring associations with primary and secondary outcomes; variables with p < 0.10 or clinical relevance were entered into multivariate models. Third, binary logistic regression was applied fOR, (a) presence of ≥ 1 complication and (b) the length of the PACU stay > 1 hour, reporting odds ratios (OR) with 95% CI. For clinical interpretability, age was modeled per decade (10-year increments) and surgical duration in 30-minute blocks; campus (Observatorio/Santa Fe) was included as a fixed effect. Linearity of continuous predictors was verified using partial residuals and LOESS curves, applying transformations or categorizations when necessary; collinearity was assessed with VIF < 5. Model performance was evaluated using area under the ROC curve (discrimination) and calibration (Hosmer-Lemeshow test and calibration curve). Pre-specified sensitivity analyses included stratified models by type of anesthesia and surgical specialty, exclusion of cases with missing critical data, and models for each main complication (pain, hypertension, and PONV).
Regarding missing data, when the proportion was ≤ 5%, complete-case analysis was used; if > 5%, multiple imputation by chained equations (MICE, m = 20) was applied assuming missing at random, combining estimates according to Rubin’s rules.
Analyses were conducted using R (RStudio) and StatPlus:mac Pro v5.9.92. Two-sided p < 0.05 was considered statistically significant. The protocol was approved by the Ethics/Research Committee of Centro Médico ABC, classified as minimal risk, and all information was treated confidentially and anonymized in accordance with institutional regulations.
Results
During the study period, a total of 2,025 patients meeting inclusion criteria were enrolled. General demographic characteristics are shown in table 2. Of the total, 27.9% were treated at the Observatorio campus and 72.1% at the Santa Fe campus. Sex distribution was balanced (50.7% men and 49.3% women), with a median age of 48 years. According to ASA classification, most patients were ASA I (30.4%) and ASA II (56.2%).
Table 2. PACU complications (n = 2,025). n (%) are shown for each event
| Variable | Summary |
|---|---|
| Shivering | Yes: 13 (0.6%). No: 2,012 (99.4%) |
| History of PONV | Yes: 11 (0.5%). No: 2,014 (99.5%) |
| PONV | Yes: 35 (1.7%). No: 1,990 (98.3%) |
| PONV Tx | Yes: 35 (1.7%). No: 1,990 (98.3%) |
| Hypotension | Yes: 20 (1.0%). No: 2,005 (99.0%) |
| Hypertension Tx | Yes: 18 (0.9%). No: 2,007 (99.1%) |
| Hypertension | Yes: 289 (14.3%). No: 1,736 (85.7%) |
| Hypertension Tx | Yes: 38 (1.9%). No: 1,987 (98.1%) |
| Unstable bradycardia | Yes: 8 (0.4%). No: 2,017 (99.6%) |
| Tachycardia | Yes: 17 (0.8%). No: 2,008 (99.2%) |
| PACU time > 1 hour | Yes: 473 (23.4%). No: 1,552 (76.6%) |
| ICU transfer | Yes: 31 (1.5%). No: 1,994 (98.5%) |
| Delirium | Yes: 9 (0.4%). No: 2,016 (99.6%) |
| Anaphylactic reaction | Yes: 2 (0.1%). No: 2,023 (99.9%) |
| Myocardial infarction | Yes: 0 (0.0%). No: 2,025 (100.0%) |
| Death | Yes: 0 (0.0%). No: 2,025 (100.0%) |
| Respiratory depression | Yes: 5 (0.2%). No: 2,020 (99.8%) |
| Neuromuscular reversal | Yes: 609 (30.1%). No: 1,416 (69.9%) |
| Opioid reversal | Yes: 2 (0.1%). No: 2,023 (99.9%) |
| Midazolam reversal | Yes: 3 (0.1%). No: 2,022 (99.9%) |
| Pain | Yes: 437 (21.6%). No: 1,588 (78.4%) |
| Urinary retention | Yes: 0 (0.0%). No: 2,025 (100.0%) |
| Incidents: in the operating room or recovery (cardiac arrest, bronchospasm, laryngospasm, pneumothorax, etc.) | No: 2,008 (99.2%). Somnolence: 2 (0.1%). Bronchospasm/cardiopulmonary arrest/advanced CPR: 2 (0.1%). Pulseless ventricular tachycardia: 1 (0.0%). Cardiac arrest: 1 (0.0%). Pneumothorax: 1 (0.0%). No response to verbal stimuli: 1 (0.0%). ICU transfer: 1 (0.0%). Pulseless electrical activity: 1 (0.0%). Laryngospasm at extubation: 1 (0.0%). Superficial and deep venous thrombosis in lower limb (common iliac), transferred to intermediate care: 1 (0.0%). Desaturation, inadvertent extubation during emergence: 1 (0.0%). Laryngospasm: 1 (0.0%). ICU/ED transfer: 1 (0.0%). Bronchospasm: 1 (0.0%). Atrial fibrillation, treated with amiodarone: 1 (0.0%) |
|
PONV: postoperative nausea and vomiting; CPR: cardiopulmonary resuscitation; Tx: treatment; PACU: post-anesthesia care unit; ICU: intensive care unit. | |
Mean surgical duration was 159 minutes, and 93.2% were elective procedures. The most frequent surgical specialties were general surgery (34.2%), trauma and orthopedics (22.1%), and urology (13.9%), whereas maxillofacial surgery represented only 0.1%. The most widely used anesthetic technique was balanced general anesthesia (86.5%), followed by total intravenous anesthesia (TIVA) (8.4%).
At least 1 comorbidity was present in 40.1% of patients, most commonly hypertension and neoplasms. In the immediate postoperative period, 45.2% experienced at least 1 complication, with pain (21.6%) and hypertension (14.3%) being the most prevalent (Table 3). No statistically significant differences were found between type of anesthesia and PACU stay longer than one hour, nor in the rate of complications during that time.
Table 3. Adjusted ORs from the regularized logistic model for PACU complication
| Predictor | Adjusted OR | 95% CI (bootstrap) | Significant |
|---|---|---|---|
| Sex: male | 1.07 | 0.89-1.35 | No |
| ASA: collapsed_II | 1.28 | 1.06-1.55 | Yes |
| ASA: collapsed_III+ | 1.84 | 1.30-2.65 | Yes |
| Type of surgery: cx_general surgery | 1.23 | 0.94-1.56 | No |
| Type of surgery: cx_gynecology | 1.29 | 0.57-2.64 | No |
| Type of surgery: cx_maxillofacial | 0.99 | 0.41-2.09 | No |
| Type of surgery: cx_neurosurgery | 0.74 | 0.50-1.10 | No |
| Type of surgery: cx_oncologic surgery | 0.71 | 0.45-1.04 | No |
| Type of surgery: cx_otolaryngology | 0.57 | 0.42-0.78 | Yes |
| Type of surgery: cx_plastic surgery | 0.63 | 0.43-0.90 | Yes |
| Type of surgery: cx_thoracic | 3.96 | 2.74-5.38 | Yes |
| Type of surgery: cx_trauma and orthopedics | 0.73 | 0.58-0.93 | Yes |
| Type of surgery: cx_urology | 0.63 | 0.47-0.85 | Yes |
| Type of surgery: cx_vascular | 1.27 | 0.66-2.82 | No |
| Technique: regional anesthesia | 1.74 | 0.64-3.81 | No |
| Technique: combined | 0.72 | 0.47-1.13 | No |
| Technique: neuraxial | 2.70 | 1.15-5.24 | Yes |
| Technique: TIVA | 0.56 | 0.37–0.90 | Yes |
| Condition: urgent | 1.32 | 0.94–2.04 | No |
| Age ≥ 65 years | 1.12 | 0.90–1.38 | No |
| Surgical time > 3 h | 1.21 | 0.94–1.56 | No |
|
* Categorical variables coded with reference categories: female, ASA I, general surgery, balanced general anesthesia, elective. OR, 95% CI (bootstrap, 200 replicates), and significance (95% CI excluding 1.0) are shown. ASA: American Society of Anesthesiologists classification; cx: surgery; 95% CI: 95% confidence interval; OR: odds ratio; PACU: post-anesthesia care unit; TIVA: total intravenous anesthesia. | |||
Figure 1 illustrates that the most frequent complications were reversal of neuromuscular blockade (30.1%), PACU stay > 1 hour (23.4%), and pain (21.6%). Hypertension reached 14.3%, while other events (eg, PONV 1.7%, antihypertensive treatment 1.9%, ICU transfer 1.5%, hypotension 1.0%, tachycardia 0.8%) were rare.
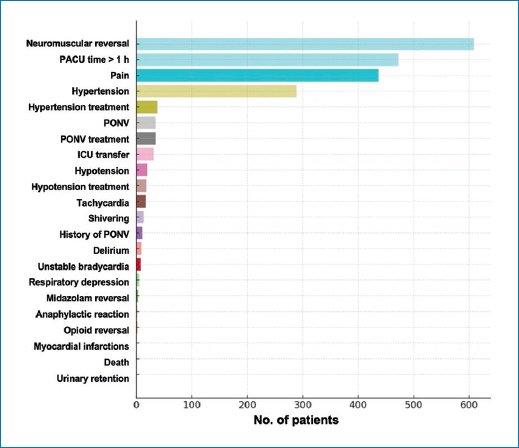
Figure 1. Frequency of complications in the PACU (excluding the most frequent category). Horizontal bars for each event; the number of patients per complication is shown. Percentages correspond to the total cohort (n = 2,025). PONV: postoperative nausea and vomiting; PACU: post-anesthesia care unit; ICU: intensive care unit.
Logistic regression model results
To estimate the risk of overall PACU complications, a binary logistic regression model was constructed with “any complication” as the outcome and pre-specified predictors: age ≥ 65 years, surgical time > 3 hours, sex, ASA (I, II, III+), type of surgery, anesthetic technique, and elective/urgent condition. These predictors were selected based on clinical plausibility and routine perioperative stratification use. Categorical variables were coded using one-hot encoding, with general surgery, balanced general anesthesia, ASA I, female sex, and elective condition as references. To reduce instability due to collinearity or sparse cells, L2 regularization was applied, and 95% CIs were obtained via bootstrap (200 replicates), which provides more robust intervals when resampling the cohort.
The model demonstrated moderate discrimination (area under the curve [AUC] = 0.631) and a Brier score of 0.22 (low-to-moderate probability error); the ROC curve is shown in figure 2. In the adjusted analysis (Table 3, Fig. 3), baseline severity and the procedural context dominated the association with the outcome. Compared with ASA I, ASA II was associated with higher risk (OR, 1.28; 95% CI, 1.06-1.55), and ASA III+ showed an additional increase (OR, 1.84; 95% CI, 1.30-2.65), forming a clinically coherent risk gradient. By specialty, thoracic surgery showed a marked increase vs general surgery (OR, 3.96; 95% CI, 2.74-5.38), whereas otolaryngology (OR, 0.57; 95% CI, 0.42-0.78), urology (OR, 0.63; 95% CI, 0.47-0.85), plastic surgery (OR, 0.63; 95% CI, 0.43-0.90), and trauma/orthopedics (OR, 0.73; 95% CI, 0.58-0.93) were associated with a lower probability of complication. Regarding anesthetic technique, neuraxial anesthesia showed higher risk (OR, 2.70; 95% CI, 1.15-5.24) and TIVA lower risk (OR, 0.56; 95% CI, 0.37-0.90) vs balanced general anesthesia. In contrast, age ≥ 65 years and surgical time > 3 hours did not demonstrate a robust independent effect after adjustment (95% CIs included 1.0), and neither sex nor urgent condition showed consistent associations in this framework. This pattern suggests that immediate risk burden primarily reflects patient complexity (ASA) and procedural characteristics (specialty and technique), rather than simple dichotomous thresholds.
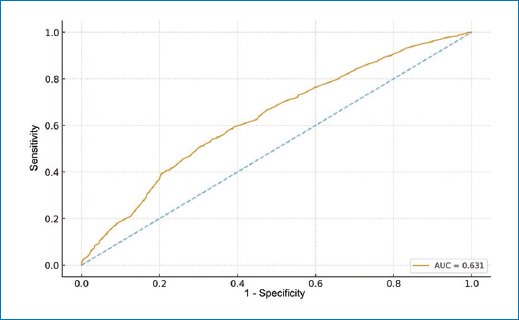
Figure 2. ROC curve of the regularized logistic model for PACU complication. The ROC curve is shown with its overall AUC (diagonal line as reference for chance). AUC: area under the curve; ROC: receiver operating characteristic; PACU: post-anesthesia care unit.
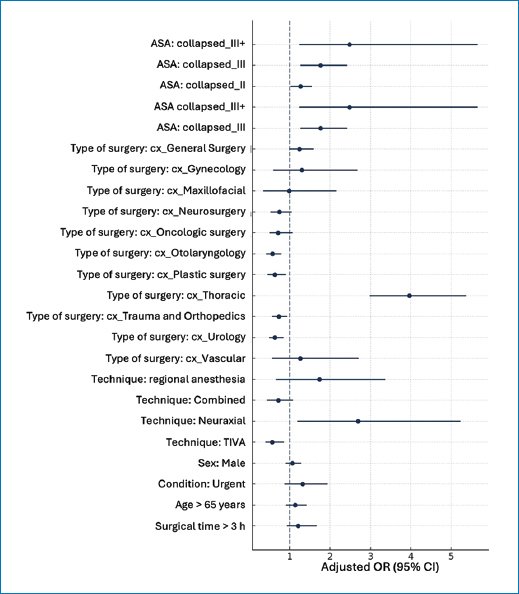
Figure 3. Forest plot of adjusted ORs and their 95% confidence intervals (bootstrap) from the regularized logistic model for complication in the post-anesthesia care unit. References: female, ASA I, general surgery, balanced general anesthesia, elective. The dashed line marks OR = 1. ASA: American Society of Anesthesiologists classification; OR: odds ratio; TIVA: total intravenous anesthesia.
Additionally, the effect of surgical time was explored as a continuous variable to capture gradual increments. In univariate logistic analysis, each additional 30 minutes was associated with an OR of 1.04 (95% CI, 1.00-1.07); in the adjusted model (same covariates), the OR per 30 minutes was 1.05 (95% CI, 1.01-1.09). These values indicate a progressive and independent increase in risk across clinically interpretable time units. The test for nonlinearity using a quadratic term did not reach statistical significance (p = .058), supporting an approximately linear relationship within the observed range; therefore, reporting the effect per 30 minutes provides a stable and clinically useful measure for decision-making. Consistently, incidence by quartiles of duration showed a growing gradient of complications from Q1 (shortest procedures) to Q4 (longest). Overall, these findings indicate that operative duration, when treated continuously, is associated with a progressive and independent increase in immediate postoperative risk, whereas the dichotomous cutoff of “> 3 hours” may underestimate this relationship.
Overall, these results show that the risk of experiencing at least 1 PACU complication is primarily determined by baseline severity and procedural context, with a gradient across ASA categories (vs ASA I: ASA II, OR, 1.28; 95% CI, 1.06-1.55; ASA III+; OR, 1.84; 95% CI, 1.30-2.65), a marked increase in thoracic surgery (OR, 3.96; 95% CI, 2.74-5.38), and lower probability in otolaryngology, urology, plastic surgery, and trauma/orthopedics (OR < 1).
Regarding the anesthetic technique, neuraxial blockade was associated with higher risk (OR, 2.70; 95% CI, 1.15-5.24) and TIVA with lower risk (OR, 0.56; 95% CI, 0.37-0.90) vs balanced general anesthesia. Surgical duration, modeled continuously, was associated with a progressive and independent increase in risk (OR per 30 min: 1.05; 95% CI, 1.01-1.09), without evidence of relevant curvature (p = 0.058), whereas the > 3-hour cutoff did not show a robust independent effect, suggesting that dichotomous thresholds may underestimate the temporal gradient. From an operational perspective, these findings support stratification and anticipatory triage in the PACU (eg, increased monitoring and resources for patients with higher ASA classification, thoracic procedures, neuraxial anesthesia, and prolonged duration), and guide process-based interventions (multimodal analgesia, hemodynamic targets, prevention of PONV and respiratory insufficiency).
Discussion
The PACU is a critical transition area where respiratory, hemodynamic, and analgesia-related risks converge, and whose safety depends on structured monitoring and standardized protocols: ASA standards require continuous surveillance of oxygenation, ventilation, circulation, neurologic status, and temperature, with pulse oximetry during the initial phase and formal patient handoff/transfer documentation upon arrival10,11.
Across different settings, the overall burden of complications can be high (eg, 54.8% in a low-resource Ethiopian cohort), reinforcing the need for clear operational definitions and reproducible processes12. From a pathophysiological standpoint, 2 frequent drivers of PACU events are atelectasis – present in 85%-90% of adults after general anesthesia and a major cause of early hypoxemia – and residual neuromuscular blockade (TOF < 0.9), whose incidence rate remains common when quantitative monitoring and reversal are not used (series reporting ~22% and up to > 50%)3,4. Added to this is PONV, with a baseline population risk of approximately 20-30% but highly preventable through multimodal prophylaxis per the 2020 consensus, which can shift the hierarchy of events toward pain and hemodynamic disturbances when prophylaxis is applied systematically. Overall, this framework supports why our study adopted sensitive thresholds to detect adequate pain control and operational hemodynamic targets13–15.
Our observational study confirmed that the frequency of PACU complications is substantial, with nearly half of patients experiencing at least one event, mainly pain and hypertension. Multivariable analysis showed that the only independent predictors were age and surgical duration, with an approximate 10% increase in risk per decade of life and 6% per additional 30 minutes of surgery. Although these magnitudes may seem modest, their cumulative effect is clinically relevant and necessitates intensified surveillance in older patients and in prolonged procedures. In contrast, no significant association was identified with anesthetic technique or with length of stay > 1 hour, likely due to homogeneity in institutional practice: predominance of balanced general anesthesia and standardized prophylaxis and monitoring protocols across both campuses. These findings reinforce that the true safety differential lies not in the isolated technique, but in the consistent execution of universal prevention and anesthetic surveillance measures.
Data suggest that temporal exposure to the surgical act and age-related vulnerability increase the risk of “any event” in a diffuse manner, rather than predisposing to a specific complication.
To place these results in context, our predominant pattern – pain and hypertension with low PONV – is consistent with frequently reported PACU events in recent reviews (pain, hemodynamic alterations, hypo-/hypertension, respiratory events, and PONV), but differs in rank order: in our setting, PONV appears controlled, likely due to systematic prophylaxis, whereas hypertension carries greater weight. In addition, the use of operational thresholds of ± 20% relative to baseline blood pressure and standardized surveillance (serial vital signs, capnography, pulse oximetry) is consistent with safety recommendations linking this approach to lower morbidity and timely event detection in the PACU; in fact, these clinical practice guidelines propose maintaining heart rate/blood pressure within 20% of baseline14,16.
In sum, we align with the expected catalog of complications, but our profile suggests that well-implemented monitoring and prophylaxis protocols can shift the burden toward pain/hypertension rather than toward PONV, reinforcing the priority of universal analgesia and blood pressure control bundles in older patients or those undergoing longer surgical procedures.
Compared with systematic reviews, where critical respiratory events in the PACU range from 0.8% to 6.9% and the most consistent predictors include surgical site and duration, urgency, and patient factors such as age and ASA – with physiological emphasis on atelectasis in 85-90% of anesthetized adults, decreased heart rate, and residual neuromuscular blockade in 33-64% upon arrival – our cohort showed a low respiratory burden. Our findings regarding age and surgical time as determinants of overall risk align with the respiratory literature, whereas the lower rate of hypoxemia/desaturation likely reflects local practice (prophylaxis, selective continuous positive airway pressure/noninvasive ventilation) rather than absence of intrinsic risk16,17.
Abebe et al.12 reported an overall incidence rate of 54.8% with respiratory and PONV predominance (22.1%) and identified female sex (aOR ≈ 2.57), anesthetic duration > 4 h (aOR ≈ 5.41), and complicated intraoperative course (aOR ≈ 2.24) as predictors; our cohort (tertiary center, more standardized protocols) showed a lower PONV burden (1.7%) and a profile dominated by pain (21.6%) and hypertension (≈14-15%). We concur on the role of temporal exposure (duration) and age as risk markers, but we did not identify an independent effect of sex or anesthetic technique after adjustment. These discrepancies are plausible given context (resource-limited setting vs high-complexity center) and protocols; the authors report lack of a checklist/discharge criteria and nonstandardized analgesia, which may increase PONV and respiratory events. Their data support strengthening checklists and clinical pathways; ours prioritize universal analgesia and blood pressure control bundles scaled by age and surgical time16.
Ma et al.10 developed a multilabel model (16,838 patients) that simultaneously predicts 6 PACU complications and shows meaningful co-occurrence, with hemodynamic instability as a central node; the highest-weight variables were age, sex, body mass index, anesthetic duration, and postoperative analgesia, with AUC, 0.69-0.93. Our study, smaller but prospective, used logistic regression for a composite outcome and found age and duration as the only independent predictors. Convergence on “age/duration” supports their use for simple bedside stratification, while the multilabel approach suggests a potential improvement for us: modeling co-occurrence (eg, pain↔hypertension) and calibrating prediction by event type to better target PACU resources11.
These results have direct practical implications. In older patients or after long surgical procedures, it is advisable to intensify standardized measures: operational targets of VAS < 4 with protocolized multimodal analgesia, clear hemodynamic thresholds with short-acting agents, monitoring of oxygenation and temperature with reproducible criteria, and a checklist confirming TOF ≥ 0.9 before operating room discharge. Because anesthetic technique showed no independent effect, the improvement focus should be on consistent execution of these bundles rather than changing technique15,17.
Interpretation warrants caution due to design and context limitations. The analyzed period overlapped with operational changes in the first half of 2020; some definitions (delirium, PONV) depend on intensity of ascertainment and treatment; and exposure to analgesic/antiemetic prophylaxis was not uniformly quantified, which may mediate associations. Although the models showed adequate discrimination and calibration, residual confounding likely persists. Future evaluations should model each complication specifically (pain, hypertension, PONV) and incorporate prophylaxis exposure as a covariate15,18.
Age and duration are consistent PACU risk markers; standardizing analgesia, blood pressure control, and neuromuscular safety bundles – with escalation along these two simple axes – is the most reasonable route to reduce events and improve post-anesthetic care quality.
Conclusions
This study identified pain and hypertension as the most frequent complications in the PACU, and no significant association was found between anesthetic type and the occurrence of adverse events. Advanced age and longer surgical duration were significantly associated with a higher risk of postoperative complications, highlighting the need for individualized surveillance and treatment strategies in these patients.
From a clinical perspective, these findings support implementation of multimodal analgesia protocols and proactive hemodynamic monitoring, especially in patients with identified risk factors. In addition, future research could focus on evaluating the impact of systematic use of regional techniques, intravenous opioids, and multimodal analgesia strategies on the incidence of post-anesthetic complications. Continuous monitoring of these events should be integrated as an essential component of anesthesiology quality improvement systems.
Funding
The authors declare that they have not received funding.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical considerations
Protection of human subjects and animals. The authors declare that no experiments on humans or animals were performed for this research.
Confidentiality, informed consent, and ethical approval. The authors have obtained approval from the Ethics Committee for the analysis of routinely collected and anonymized clinical data; therefore, individual informed consent was not required. Relevant ethical recommendations have been followed.
Declaration on the use of artificial intelligence. The authors declare that no generative artificial intelligence was used in the writing or creation of the content of this manuscript.
