Introduction
Prostate cancer is the 2nd most frequent malignant neoplasm among men worldwide. In 2022, the International Agency for Research on Cancer reported 10,311,610 cases globally, with an incidence rate of 14.2% and a mortality rate of 9.6%. In Mexico, of 25,000 reported cases, 7,500 resulted in death1,2.
Early detection impacts prognosis; therefore, measurement of prostate-specific antigen (PSA) is recommended starting at 40 years of age and digital rectal examination beginning at 55 years of age3,4. Clinical signs are absent in most cases. Symptomatic patients may present with erectile dysfunction, decreased libido, nocturia, urinary urgency, urinary frequency, urinary tract infection, hematospermia, and perineal or scrotal pain. Patients with metastatic disease may present with bone pain and deterioration of general condition5.
Castration-resistant prostate cancer
This type of prostate cancer exhibits resistance to prior surgical and/or medical therapy, requiring the use of new drugs to reduce PSA and testosterone levels to halt disease progression.
Diagnosis requires the following characteristics5,6:
- –Low serum testosterone concentrations after castration < 50 ng/mL (1.7 mmol/L).
- –3 consecutive PSA elevations at 1-week intervals, with 2 increases of 50% or PSA > 2 ng/mL (biochemical progression).
- –≥ 2 bone lesions on bone scintigraphy or nonbone metastases according to RECIST criteria, even with castration-level testosterone (< 50 ng/mL) (radiologic progression)5,6.
- –However, despite new treatments and mechanisms of action, there remains no substantial impact on prognosis in patients with this type of cancer7,8. The development of metastases represents a crucial event in disease progression, as the onset of castration resistance reflects a potentially lethal condition9.
Pathophysiology of castration-resistant prostate cancer
Several mechanisms may explain tumor cell proliferation despite low circulating testosterone concentrations:
- –Mutations in the androgen receptor (AR), leading to a hypersensitive receptor and gene rearrangements10.
- –Splice variant 7 of the androgen receptor (AR-V7 isoform), resulting in constitutive activation and stimulation of tumor growth11.
- –De novo intratumoral androgen synthesis mediated by enzymes expressed in tumor tissue12.
- –Effects of microRNAs13.
Abiraterone acetate
Abiraterone acetate is a selective androgen biosynthesis inhibitor. It is a potent, selective, and irreversible inhibitor of androgen biosynthesis through permanent blockade of 17α-hydroxylase (CYP17), a crucial enzyme in androgen synthesis14.
Enzalutamide
Enzalutamide directly inhibits the androgen receptor (AR), blocking androgen binding to the receptor, inhibiting nuclear translocation of the activated AR, and preventing recruitment of coactivators15.
Methods
Study design
We conducted an observational, descriptive, retrospective, cross-sectional study was conducted.
Study population
Databases of antiandrogen medications available at the participating medical unit were reviewed to identify patients receiving treatment who met inclusion criteria.
Patients aged 18 years or older with a diagnosis of prostate cancer, treated with abiraterone and enzalutamide, and with complete health records were included. Data were entered into an Excel database and subsequently analyzed using SPSS version 26 for Windows.
Variables included age, Eastern Cooperative Oncology Group (ECOG) performance status, clinical stage, Gleason score, histopathologic findings, metastases (site and number), initial PSA level, initial testosterone level, most recent PSA level at follow-up, treatment drug, and progression-free survival (PFS).
Statistical analysis
Descriptive statistics were used, including measures of central tendency, dispersion, frequencies, and proportions. Survival curves were constructed.
Results
A total sample of 63 patients was obtained.
The mean age was 72 years, with a mode of 80 and a median of 73. Regarding decades of life, most patients were in their 8th decade, accounting for 29 (46%) individuals; the remaining results are shown in table 1.
Table 1. Decades of life
| Decade | n | % |
|---|---|---|
| Eighth | 29 | 46% |
| Seventh | 21 | 33% |
| Ninth | 7 | 11% |
| Sixth | 5 | 8% |
| Fifth | 1 | 2% |
| Total | 63 | 100% |
|
n: number of patients per decade; %: percentage of patients per decade. | ||
Of the 63 patients, 10 (16%) had an ECOG performance status of 0, 37 (59%) ECOG 1, and 10 (16%) ECOG 2; the remaining patients were classified as ECOG 3 or 4.
Regarding staging, for practical purposes the clinical stages used in this study ranged only from I to IV, eliminating the subcategories of stage II (IIA and IIB). In this cohort, 53 (84%) patients were diagnosed with stage IV disease, 5 (8%) with stage III, 4 (6%) with stage II, and 1 (2%) with stage I.
With respect to histology, all patients had prostatic acinar adenocarcinoma. Of these, 8 (13%) showed neuroendocrine differentiation. Regarding the Gleason score, which reflects tumor cell differentiation, 23 (36.5%) patients had a Gleason score of 9, consistent with poorly differentiated neoplasms and worse prognosis; only 1 (1.6%) patient had a Gleason score of 4, considered indicative of favorable prognosis. The remaining results are shown in table 2.
Table 2. Gleason score by patients
| Gleason | n | % |
|---|---|---|
| 4 | 1 | 1.6% |
| 6 | 3 | 4.8% |
| 7 | 14 | 22.2% |
| 8 | 19 | 30.2% |
| 9 | 23 | 36.5% |
| 10 | 3 | 4.8% |
| Total | 63 | 100% |
|
n: number of patients; %: percentage. | ||
Primary site of metastasis
All patients had bone as the primary metastatic site. However, 57 (90.5%) had bone as the only metastatic site, 2 (3.6%) had 2 metastatic sites, and 4 (6.4%) had 3 metastatic sites. The distribution of primary metastatic sites is shown in table 3.
Table 3. Number of metastases
| Metastasis sites | n | % |
|---|---|---|
| Bone | 57 | 90.5% |
| Bone, retroperitoneal | 2 | 3.1% |
| Bone, retroperitoneal, and para-aortic | 1 | 1.6% |
| Bone, retroperitoneal, and mesenteric | 1 | 1.6% |
| Bone, lung, and retroperitoneal | 1 | 1.6% |
| Bone, lung, and liver | 1 | 1.6% |
| Total | 63 | 100% |
|
n: number of patients; %: percentage. | ||
Treatment
Of the 63 patients included in the study, 36 (57%) received enzalutamide at a standard dose of 160 mg daily, and 27 (43%) received abiraterone at a standard dose of 1,000 mg daily. At the time of analysis, 38 (60.32%) patients had not experienced disease progression. Among the 25 (39.68%) patients with progression, 12 (48%) had a single type of progression (radiologic or biochemical), and 13 (52%) > 1 type (both biochemical and radiologic progression).
Treatment with abiraterone
Among the 27 (43%) patients treated with abiraterone, 16 (59%) had not received prior chemotherapy (pre-docetaxel), whereas 11 (41%) had received chemotherapy before abiraterone administration (post-docetaxel). Patients from the pre-docetaxel group achieved a mean progression-free survival (PFS) of 17.938 months; 80% reached PFS at 10 months, gradually decreasing to 20% at 26 months. Minimum survival was 4 months and maximum survival was 37 months. Patients from the post-docetaxel group achieved a mean PFS of 15 months; 80% reached PFS at 6 months, gradually decreasing to 20% at 25 months. Minimum survival was 4 months and maximum survival was 33 months. PFS results for both groups (pre- and post-docetaxel) are shown in table 4 and figure 1.
Table 4. Progression-free survival in patients treated with abiraterone
| CT | Mean | Median | ||||||
|---|---|---|---|---|---|---|---|---|
| 95% CI | 95% CI | |||||||
| Estimate (months) | Std. error | Lower limit | Upper limit | Estimate (months) | Std. error | Lower limit | Upper limit | |
| Pre | 17.938 | 2.323 | 13.385 | 22.490 | 18.000 | 2.667 | 12.773 | 23.227 |
| Post | 15.000 | 2.883 | 9.350 | 20.650 | 12.000 | 1.651 | 8.763 | 15.237 |
| Overall | 16.741 | 1.796 | 13.220 | 20.262 | 15.000 | 3.894 | 7.367 | 22.633 |
|
Post: post-docetaxel; Pre: pre-docetaxel; CT: chemotherapy; CI: confidence interval. | ||||||||
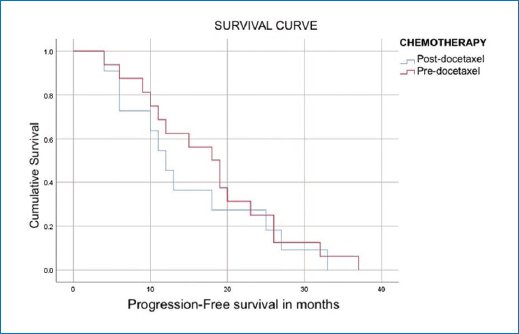
Figure 1. Progression-free survival in patients treated with abiraterone in the pre- and post-docetaxel settings.
Treatment with enzalutamide
Of the 36 patients treated with enzalutamide, 23 (63.8%) had not received prior chemotherapy (pre-docetaxel), and 13 (36.2%) had received chemotherapy before enzalutamide administration (post-docetaxel). Patients from the pre-docetaxel group achieved a mean PFS of 19.739 months; 80% reached PFS at 7 months, gradually decreasing to 20% at 35 months. Minimum survival was 3 months and maximum survival was 46 months. Patients from the post-docetaxel group achieved a mean PFS of 21.077 months; 80% reached PFS at 11 months, gradually decreasing to 20% at 33 months. Minimum survival was 8 months and maximum survival was 36 months. PFS results for both groups (pre- and post-docetaxel) are shown in table 5 and figure 2.
Table 5. Progression-free survival in patients treated with enzalutamide
| CT | Mean | Median | ||||||
|---|---|---|---|---|---|---|---|---|
| 95% CI | 95% CI | |||||||
| Estimate (months) | Std. error | Lower limit | Upper limit | Estimate (months) | Std. error | Lower limit | Upper limit | |
| Pre | 19.739 | 2.796 | 14.258 | 25.220 | 15.000 | 4.791 | 5.609 | 24.391 |
| Post | 21.077 | 2.914 | 15.365 | 26.789 | 17.000 | 3.595 | 9.954 | 24.046 |
| Overall | 20.222 | 2.050 | 16.204 | 24.241 | 17.000 | 1.491 | 14.078 | 19.922 |
|
Post: post-docetaxel; Pre: pre-docetaxel; CT: chemotherapy; CI: confidence interval. | ||||||||
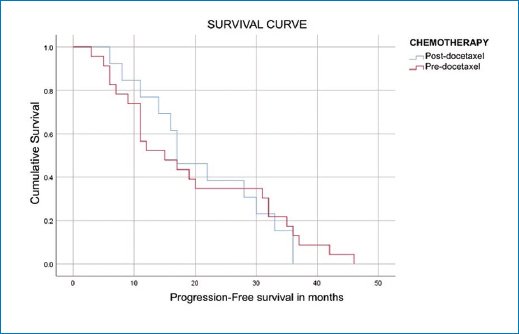
Figure 2. Survival in patients treated with enzalutamide in the pre- and post-docetaxel settings.
Discussion
In the clinical trials evaluating abiraterone, namely COU-AA-302 (pre-docetaxel) and COU-AA-301 (post-docetaxel), survival outcomes were assessed in patients with prostate cancer treated with this agent. In the COU-AA-302 trial, the median radiographic progression-free survival (rPFS) was 16.5 months; in the COU-AA-301 trial, the median rPFS was 5.6 months9,16,17.
In the present analysis, patients from the pre-docetaxel group achieved a PFS of 17.938 months, whereas those from the post-docetaxel group achieved a PFS of 15.0 months.
Similarly, in the enzalutamide trials PREVAIL (pre-docetaxel) and AFFIRM (post-docetaxel), survival outcomes were evaluated in patients with prostate cancer receiving this treatment. In the PREVAIL trial, the median rPFS was 20.0 months; in the AFFIRM trial, the median rPFS was 16.7 months15,18–20.
In the present study, patients from the pre-docetaxel group achieved a PFS of 19.739 months, while those from the post-docetaxel group achieved a PFS of 21.077 months.
Conclusions
PFS in patients with castration-resistant prostate cancer in the studied population was higher than that reported in international trials, with the exception of the pre-docetaxel group treated with enzalutamide, which showed similar results.
Funding
The authors declare that they have not received funding.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical considerations
Protection of human subjects and animal. The authors declare that the procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation and with the World Medical Association and the Declaration of Helsinki. The procedures were authorized by the Institutional Ethics Committee.
Confidentiality, informed consent, and ethical approval. The authors have obtained approval from the Ethics Committee for the analysis of routinely collected and anonymized clinical data; therefore, individual informed consent was not required. Relevant ethical recommendations have been followed.
Declaration on the use of artificial intelligence. The authors declare that no generative artificial intelligence was used in the writing or creation of the content of this manuscript.
